23 March 2018: Spring Expo
Begin the new season as you mean to go on! Whether you are looking to build your network, promote your business, find new suppliers or
6-13 March: China Life Sciences Market Access Roadshow 2018
In the context of progressive change in the Chinese life science market, and a growing interest from UK companies, the UK Department for International Trade
Importance of emergency treatment of acute ocular surface acid attacks using amniotic membrane
Andrew Hopkinson, CEO of NuVision, has written a blog post discussing the importance of effective treatment in the 72 hours following an ocular surface acid attack.

Why Use Error Is Everyone’s Responsibility
Dr Sinziana M Popescu, a Research Fellow at the Medical Devices Testing and Evaluation Centre (MD-TEC), has penned an article within the latest edition of

Association of British Healthcare Industries (ABHI) Healthy Outside the EU: Update
ABHI has recently published a document, serving as an update to their April 2017 piece ‘Healthy outside the EU – Strategy for a thriving MedTech industry.’

Medical Devices Testing and Evaluation Centre (MD-TEC) Opens in Birmingham
The new Medical Devices Testing and Evaluation Centre (MD-TEC) had its grand opening on 17/01/2018. The event, held at the Institute of Translational Medicine (ITM),
Tech-talk: MTI meets Tom Clutton-Brock
Dr Tom Clutton-Brock is the clinical director of the new Medical Devices Testing and Evaluation Centre (MD-TEC), based in the Institute of Translational Medicine (ITM)
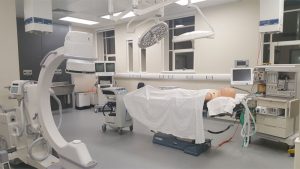
MD-TEC Launch
An event to mark the launch of The Medical Devices Testing and Evaluation Centre (MD-TEC) is due to take place on January 17th, 2018. The
NIHR Surgical MedTech Co-operative Launch Event 01/03/2018
In January 2018 the NIHR Colorectal Therapies Healthcare Technologies Co-operative will become the NIHR Surgical MedTech Co-operative. The new Surgical MedTech Co-operative will build a

NIHR Trauma Management MedTech Co-operative Launch Event 07/02/2018
As you may already know, we were delighted to have been successful with the NIHR MIC applications for the NIHR Trauma Management MedTech Co-operative which
MedTech Europe Flowcharts on New Regulatory Requirements
On September 28th, MedTech Europe published two flowcharts to help navigate the requirements of the medical devices (MD) and in vitro diagnostic (IVD) regulatory frameworks during the


